What is immunity? How do we develop it? – Part 3 – Complement System
January 13th, 2021 | by Saren Tasciyan | posted in Biology, Immunology, Science
If you didn’t read it yet, here is the part 1.
We have seen more complex parts of the immune system. Cells and their interaction creates lot’s of dynamics. Cells have many signaling and signal processing mechanisms, which can be hard to understand or analyze. In this shorter article, I will try to explain complement system.
Complement system is actually bunch of proteins in the serum (liquid in the blood), which work together to kill germs. It has the function to recognize and destroy common targets. Bacteria for instance, have many shared chemicals on their surface. For instance cell wall (animals don’t have cell walls) has a unique chemical structure. On the other hand, there is absolutely no reason of a bacteria to be present in the blood. It is ok and preferred to have bacteria in the guts, on the skin, in the mucous or even on the eyes. This is healthy. But not in the blood. Some bacteria can be sneaky and enter the blood stream. There the complement system kicks in, which launches a series of processes to label, bind and destroy the bacterium. This system also recruits immune cells for help.
There are 3 pathways (pathways in biology mean a cascade of events, e.g. a chain of proteins doing a job step by step, a number of enzymes producing a chemical step by step). Classical pathway, lectin pathway and the alternative pathway.
Let’s start with the classical pathway. You have heard about antibodies, which the B cells produce against a pathogen after an infection. Those antibodies have multiple functions but one of these functions is the complement activation via classical pathway.
- Antibody floating around bumps into it’s target by chance. This leads to antibody binding to the target. This product is called an antibody-antigen complex. (complexes are two or more molecules bound to each other)
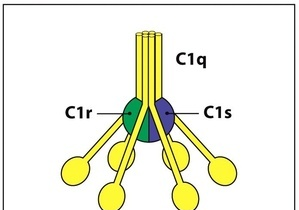
- C1 protein complex binds to the antibody-antigen complex.

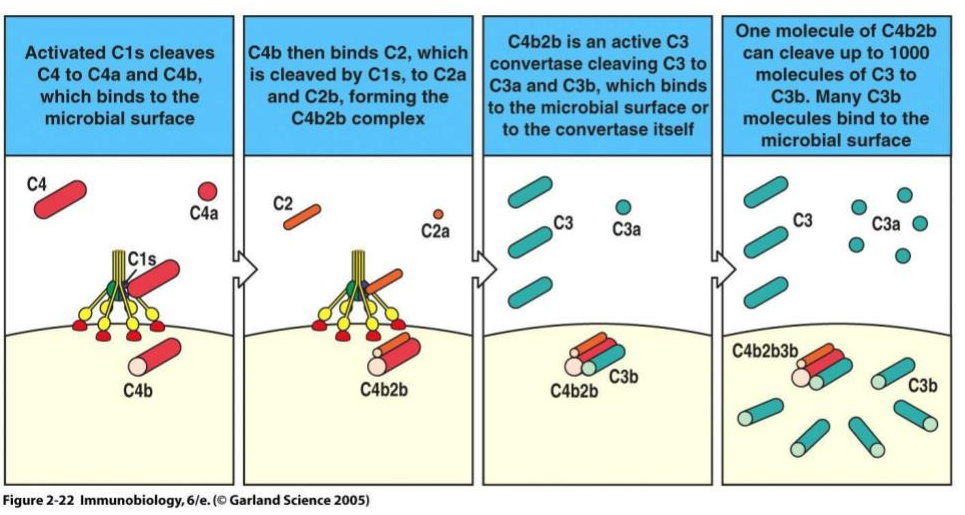
Left illustration (you don’t say captain obvious), right electron microscopy. Taken from Janeway’s immunobiology courtesy of K.B.M. Reid for the book. - C1 has enzymatic activity to cleave C4 ( 🙂 not the explosive) and C2.
- C4b (part of the C4 after cleavage, C4 cleavage :D) and C2a come together to make the C3 convertase (biochemists aren’t very creative with names “-ase” suffix means that it is an enzyme. An enzyme catalyzes chemical reactions). Also, C4b may stick to the pathogen.
- That C3 convertase converts (what a surprise) 100s-1000s of C3. Converted C3 joins the cult by also complexing and sticking to the bacterium.
- Finally, the membrane attack system (MAS) is triggered. Which involved C5, C6, C7, C8 and C9. This starts with the conversion of C3 convertase into a C5 convertase, which converts and recruits C5 to the site. Then C5-C8 gets assembled and anchored on the bacterium.
- This complex inserts bunch of C9, which makes a hole on the surface. Holes mean loss of content and influx of unwanted materials from outside. Bacteria also lose the proton gradient over their membrane. This is required to generate energy for them! Also digestive enzymes can move in to eat the bacterium from inside out. Amazing isn’t it?

I mentioned other pathways, which are similar except they are triggered by other mechanisms. They don’t require antibodies but proteins, which recognize conserved chemicals on germs’ surfaces.

On top of this, many cleaved products float away to call the rest of the immune system. For instance, C3a acts both as cytokine and chemokine, which I wrote about in the second part. This call for help tries to ensure, nothing skips the attention of the immune system.
This sounds all perfect and amazing. However, don’t forget that infection biology is a story of constant war. We do still get sick, because pathogens adapt by evolving. They try to trick our immune system. So our immune system is evolved to have redundancy. This means that if one system fails other systems can take over to finish the job.
I will end here with an amazing animation from one of my favorite YouTube channels: kurzgesagt. In the next article, I will write about a timeline of a typical viral infection.



